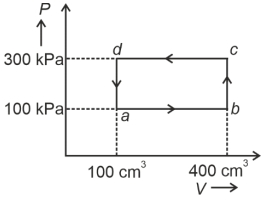
A cyclic process of a thermodynamic system is taken through $a$ $b$ $c$ $d$ $a$. The work done by the gas along the path $b$ $c$ is

NEET 2024, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
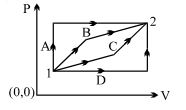
- 1An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 2View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 3Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
- 4Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperatureView Solution
- 5View SolutionWhen you make ice cubes, the entropy of water
- 6Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
- 7At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$View Solution
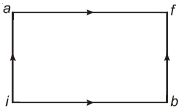
- 8When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
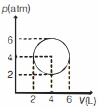
- 9An ideal gas undergoes a circular cycle centred at $4 \,atm , 4 L$ as shown in the diagram. The maximum temperature attained in this process is close toView Solution
- 10A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
