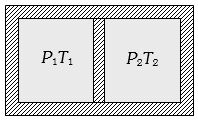
It is given that displacement of piston be in final state $x$ and if $A$ is the area of cross-section of the piston.
Hence the final volumes of the left and right part finally can be given by figure as
${V_L} = \frac{{{V_0}}}{2} + Ax$ and ${V_R} = \frac{{{V_0}}}{2} - Ax$
As it is given that the container walls and the piston are adiabatic in left side and the gas undergoes adiabatic expansion and on the right side the gas undergoes adiabatic compressive.
Thus we have for initial and final state of gas on left side
${P_1}{\left( {\frac{{{V_0}}}{2}} \right)^\gamma } = {P_f}{\left( {\frac{{{V_0}}}{2} + Ax} \right)^\gamma }$ ..... $(i)$
Similarly for gas in right side, we have ${P_2}{\left( {\frac{{{V_0}}}{2}} \right)^\gamma } = {P_f}{\left( {\frac{{{V_0}}}{2} - Ax} \right)^\gamma }$ ..... $(ii)$
From eq. $(i)$ and $(ii)$
$\frac{{{P_1}}}{{{P_2}}} = \frac{{{{\left( {\frac{{{V_0}}}{2} + Ax} \right)}^\gamma }}}{{{{\left( {\frac{{{V_0}}}{2} - Ax} \right)}^\gamma }}}$ ==> $Ax = \frac{{{V_0}}}{2}\frac{{\left[ {P_1^{1/\gamma } - P_2^{1/\gamma }} \right]}}{{\left[ {P_1^{1/\gamma } + P_2^{1/\gamma }} \right]}}$
Now from equation $(i)$
${P_f} = \frac{{{P_1}{{\left( {\frac{{{V_0}}}{2}} \right)}^\gamma }}}{{{{\left[ {\frac{{{V_0}}}{2} + Ax} \right]}^\gamma }}}$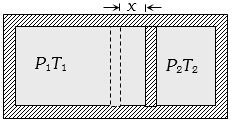
Download our appand get started for free
Similar Questions
- 1An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
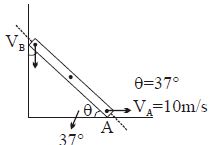
- 2Find $V_B = ?$View Solution
- 3View SolutionOut of the following which quantity does not depend on path
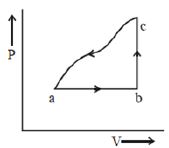
- 4A sample of an ideal gas is taken through the cyclic process $abca$ as shown in the figure. The change in the internal energy of the gas along the path $ca$ is $-180\, J$. The gas absorbs $250\, J$ of heat along the path $ab$ and $60\, J$ along the path $bc$. The work done by the gas along the path $abc$ is ..... $J$View Solution
- 5View SolutionIf a gas is heated at constant pressure, its isothermal compressibility
- 6During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
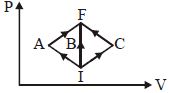
- 7In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas isView Solution
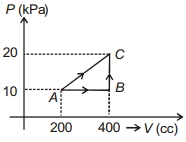
- 8If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
- 9During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
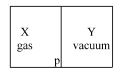
- 10A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?View Solution