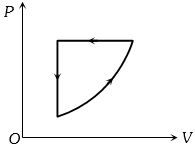
In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?
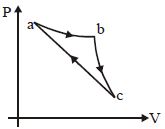

Medium
For isothermal : $\Delta U = 0$
For adiabatic : $\Delta U = -\Delta W$
For adiabatic : $\Delta U = -\Delta W$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The adiabatic elasticity of hydrogen gas $(\gamma = 1.4)$ at $NTP$ isView Solution
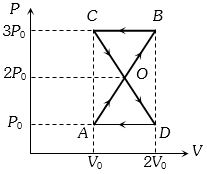
- 2A thermodynamic system undergoes cyclic process $ABCDA $ as shown in figure. The work done by the system in the cycle isView Solution
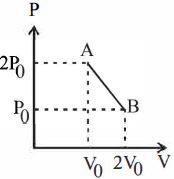
- 3'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
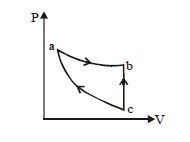
- 4An ideal gas expands isothermally along $ab$ and does $600\,J$ of work. During the processView Solution
- 5One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given byView Solution
- 6For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
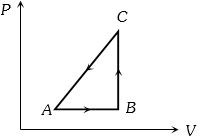
- 7The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done on the system in going from $A → B → C$ is $50 J$ and $ 20\, cal$ heat is given to the system. The change in internal energy between $A$ and $C$ is ...... $J$View Solution
- 8An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
- 9The temperature of a hypothetical gas increases to $\sqrt 2 $ times when compressed adiabatically to half the volume. Its equation can be written asView Solution
- 10A system is given $300$ calories of heat and it does $600$ joules of work. How much does the internal energy of the system change in this process ..... $J$. ($J = 4.18$ joules/cal)View Solution
