One mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure.
The change in internal energy of the gas during the transition is ............$\;kJ$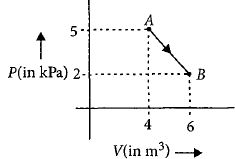
AIPMT 2015, Diffcult
We know, $\Delta U = n{C_v}\Delta T$
$ = n\left( {\frac{{5R}}{2}} \right)\left( {{T_B} - {T_A}} \right)$ $[for\,diatomic\,gas,{C_v} = \frac{{5R}}{2}]$
$ = \frac{{5nR}}{2}\left( {\frac{{{P_B}{V_B}}}{{nR}} - \frac{{{P_A}{V_A}}}{{nR}}} \right)$
$\left[ {PV = nRT} \right]$
$ = \frac{5}{2}\left( {{P_B}{V_B} - {P_A}{V_A}} \right)$
$ = \frac{5}{2}\left( {2 \times {{10}^3} \times 6 - 5 \times {{10}^3} \times 4} \right)$
$ = \frac{5}{2}\left( { - 8 \times {{10}^3}} \right) = - 20\,kJ$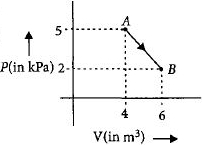
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
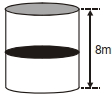
- 1A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic $($perfect thermal conductor$)$ frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top $($in $m )$ will be $. . . . . ($take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1} ).$View Solution
- 2$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 3View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 4One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
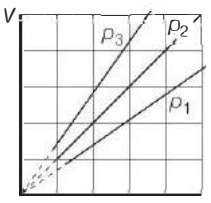
- 5View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
- 6$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
- 7For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
- 8An ideal heat engine exhausting heat at $77\,^oC$. To have a $30\%$ efficiency. It must take heat at...... $^oC$View Solution
- 9Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 10View SolutionIn isothermic process, which statement is wrong
