The slopes of isothermal and adiabatic curves are related as
Medium
(c) For Isothermal process $PV = $constant
$ \Rightarrow \left( {\frac{{dP}}{{dV}}} \right) = \frac{{ - P}}{V} = $ Slope of Isothermal curve
For adiabatic$P{V^\gamma } = $constant
$ \Rightarrow \frac{{dP}}{{dV}} = \frac{{ - \gamma P}}{V} = $ Slop of adiabatic curve slope
$ \Rightarrow \left( {\frac{{dP}}{{dV}}} \right) = \frac{{ - P}}{V} = $ Slope of Isothermal curve
For adiabatic$P{V^\gamma } = $constant
$ \Rightarrow \frac{{dP}}{{dV}} = \frac{{ - \gamma P}}{V} = $ Slop of adiabatic curve slope
Clearly, ${\left( {\frac{{dP}}{{dV}}} \right)_{{\rm{adiabatic}}}} = \gamma {\left( {\frac{{dP}}{{dV}}} \right)_{{\rm{Isothermal }}}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
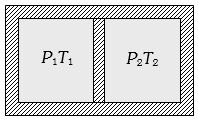
- 1Following figure shows on adiabatic cylindrical container of volume ${V_0}$ divided by an adiabatic smooth piston (area of cross-section = $A$ ) in two equal parts. An ideal gas $({C_P}/{C_V} = \gamma )$ is at pressure $P_1$ and temperature $T_1$ in left part and gas at pressure $P_2$ and temperature $T_2$ in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose $ x$ = displacement of the piston)View Solution
- 2A polyatomic gas $\left( {\gamma = \frac{4}{3}} \right)$ is compressed to $\frac{1}{8}$ of its volume adiabatically. If its initial pressure is ${P_o}$, its new pressure will beView Solution
- 3One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 4For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 5A thermally insulated container is divided into two parts by a screen. In one part the pressure and temperature are $P$ and $T$ for an ideal gas filled. In the second part it is vacuum. If now a small hole is created in the screen, then the temperature of the gas willView Solution
- 6View SolutionBy opening the door of a refrigerator placed inside a room you
- 7Helium at ${27^o}C$ has a volume of $8$ litres. It is suddenly compressed to a volume of $1$ litre. The temperature of the gas will be ....... $^oC$ $[\gamma = 5/3]$View Solution
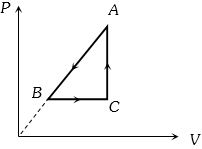
- 8$P-V$ diagram of a cyclic process $ABCA$ is as shown in figure. Choose the correct statementView Solution
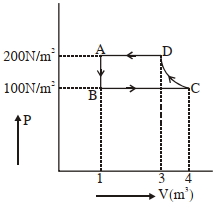
- 9The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution
- 10View SolutionA given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an
