Which of the following is a slow process
Easy
isothermal processes are necessarily slow as they require heat transfer to remain at the same temperature which is done by being in thermal equilibrium with some reservoir. $A$ process will be isothermal only if it happens on timescales larger than the timescale required for effective heat transfer.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
- 2A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 3View SolutionIn isothermal expansion, the pressure is determined by
- 4Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
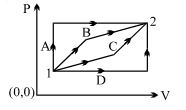
- 5An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 6View SolutionIn isothermic process, which statement is wrong
- 7A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
- 8$n-$ $moles$ of an ideal gas with constant volume heat capacity $C_v$ undergo an isobaric expansion by certain volume. The ratio of the work done in the process, to the heat supplied isView Solution
- 9In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
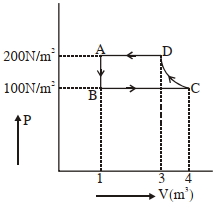
- 10The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution