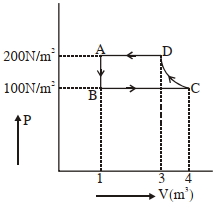
The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)

JEE MAIN 2021, Medium
Adiabatic process is from $C$ to $D$
$WD =\frac{ P _{2} V _{2}- P _{1} V _{1}}{1-\gamma}$
$=\frac{P_{D} V_{D}-P_{C} V_{C}}{1-\gamma}$
$=\frac{200(3)-(100)(4)}{1-1.4}$
$=-500\, J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 2Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
- 3For an isothermal expansion of a perfect gas, the value of $\frac{{\Delta P}}{P}$ is equalView Solution
- 4View SolutionIn Carnot engine the work done by working substance is equivalent to
- 5One mole of an ideal gas is contained with in a cylinder by a frictionless piston and is initially at temperature $T$. The pressure of the gas is kept constant while it is heated and its volume doubles. If $R$ is molar gas constant, the work done by the gas in increasing its volume isView Solution
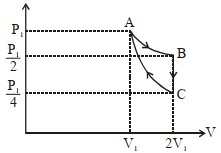
- 6If one mole of an ideal gas at $\left( P _{1}, V _{1}\right)$ is allowed to expand reversibly and isothermally ($A$ to $B$ ) its pressure is reduced to one-half of the original pressure (see $figure$). This is followed by a constant volume cooling till its pressure is reduced to one-fourth of the initial value $( B \rightarrow C ) .$ Then it is restored to its initial state by a reversible adiabatic compression ($C$ to $A$). The net workdone by the gas is equal to ...... .View Solution
- 7Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
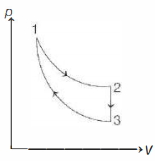
- 8The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
- 9View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
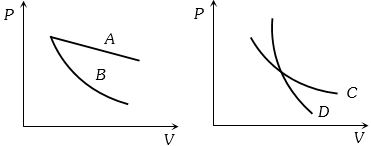
- 10In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution