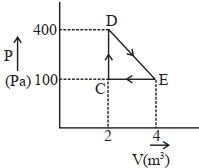
In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance is
Medium
(a) Coefficient of performance $K = \frac{{{T_2}}}{{{T_1} - {T_2}}}$
$ = \frac{{(273 - 23)}}{{(273 + 27) - (273 - 23)}}$$ = \frac{{250}}{{300 - 250}} = \frac{{250}}{{20}} = 5$
$ = \frac{{(273 - 23)}}{{(273 + 27) - (273 - 23)}}$$ = \frac{{250}}{{300 - 250}} = \frac{{250}}{{20}} = 5$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 2View SolutionWhich of the following is not a thermodynamics co-ordinate
- 3A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 4A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 5If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 6An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution
- 7If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 8View SolutionIf an ideal gas is compressed isothermally then
- 9View SolutionThe efficiency of a Carnot engine depends upon
- 10A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution