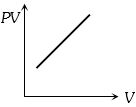
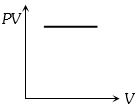
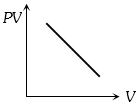
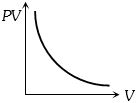
Which one the following graphs represents the behaviour of an ideal gas
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The equation of state of $n$ moles of a non-ideal gas can be approximated by the equation $\left(p+\frac{n^2 \alpha }{V^2}\right)(V-n b)=n R T$ where $a$ and $b$ are constant characteristics of the gas. Which of the following can represent the equation of a quasistatic adiabat for this gas (assume that, $C_V$ is the molar heat capacity at constant volume is independent of temperature)?View Solution
- 2An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :View Solution
- 3Two gases-argon (atomic radius $0.07 \;\mathrm{nm}$,atomic weight $40$ ) and xenon (atomic radius $0.1\; \mathrm{nm},$ atomic weight $140$ ) have the same number density and are at the same temperature. The raito of their respective mean free times is closest toView Solution
- 4On any planet, the presence of atmosphere implies (${C_{rms}}$= root mean square velocity of molecules and ${V_e}$= escape velocity)View Solution
- 5Find the ratio of specific heat at constant pressure to the specific heat constant volume for $N{H_3}$View Solution
- 6View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 7The volume of a gas will be double of what it is at $0°C$ (pressure remaining constant) atView Solution
- 8A monoatomic ideal gas of two moles is taken through a cyclic process starting from $1$ as shown. $\frac{{{V_2}}}{{{V_1}}} = 2$ and $\frac{{{V_4}}}{{{V_1}}} = 4$ and temperature iast : $1$ is $T_1 = 27\,^oC$. The temperature at $2$ i.e., $T_2$ ...... $K$View Solution
- 9One gram mole of an ideal gas $A$ with the ratio of constant pressure and constant volume specific heats $\gamma_{A}=5 / 3$ is mixed with $n$ gram moles of another ideal gas $B$ with $\gamma_{B}=7 / 5$. If the $\gamma$ for the mixture is $19 / 13$, then what will be the value of $n$ ?View Solution
- 10View SolutionThe vapour of a substance behaves as a gas