The equation of state of $n$ moles of a non-ideal gas can be approximated by the equation $\left(p+\frac{n^2 \alpha }{V^2}\right)(V-n b)=n R T$ where $a$ and $b$ are constant characteristics of the gas. Which of the following can represent the equation of a quasistatic adiabat for this gas (assume that, $C_V$ is the molar heat capacity at constant volume is independent of temperature)?
KVPY 2013, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Open container contains $x$ mole of gas at $T$ $kelvin$ If temperature is tripled, then the amount of gas exit the container isView Solution
- 2The volume occupied by the molecules contained in $4.5\,kg$ water at $STP$, if the intermolecular forces vanish away is ........ $m ^{3}$View Solution
- 3At $0°C $ the density of a fixed mass of a gas divided by pressure is $x.$ At $100°C,$ the ratio will beView Solution
- 4$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:View Solution
- 5View SolutionMoon has no atmosphere because
- 6The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]View Solution
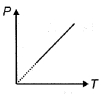
- 7At which of the following temperature would the molecules of a gas have twice the average kinetic energy they have at $20°C$View Solution
- 8The ratio of total energy of all molecules of one mole $O_2$ to the total energy of all molecules of two moles of $He$ at the same temprature isView Solution
- 9Consider a mixture of gas molecule of types $A, B$ and $C$ having masses $m_{A}\,<\,m_{B}\,<\,m_{C}$ ratio of their root mean square speeds at normal temperature and pressure is :View Solution
- 10A vessel of volume $8\, litre$ contains an ideal gas at $300\, K$ and $2\, atm$ pressure. The gas is allowed to leak till pressure become $125\, kpa$ calculate amount of moles which leak out if temperature remain constant ...... $moles$View Solution
