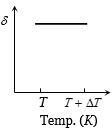
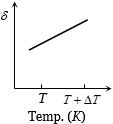
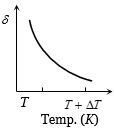
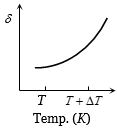
An ideal gas is initially at temperature $T$ and volume $V.$ Its volume is increased by $\Delta V$ due to an increase in temperature $\Delta T,$ pressure remaining constant. The quantity $\delta = \Delta V/(V\Delta T)$ varies with temperature as
IIT 2000, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $B$. Then the expansion of the same ideal gas of mass $2 \,\,m$ at a pressure $2P$ is given by the straight lineView Solution
- 2The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 3View SolutionAt constant volume, temperature is increased. Then
- 4Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent degree of freedom $f$ of the mixture isView Solution
- 5The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 6Two identical adiabatic vessels are filled with oxygen at pressure $P_1$ and $P_2 (P_1 > P_2).$ The vessels are interconnected with each other by a nonconducting pipe. If $U_{01}$ and $U_{02}$ denote initial internal energy of oxygen in first and second vessel respectively and $U_{f_1}$ and $U_{f_2}$ denote final internal energy values, than :View Solution
- 7$N( < 100)$ molecules of a gas have velocities $1, 2, 3\,.\,.\,.\,.\,.N/km/s$ respectively. ThenView Solution
- 8Statement$-1$ : Real gas approaches ideal gas behaviour for low pressures and high temperatures.View Solution
statement$-2 $: At low pressure, density of gas is very low. - 9If the degree of freedom of a gas are $f,$ then the ratio of two specific heats ${C_P}/{C_V}$ is given byView Solution
- 10A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)View Solution