Two gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA measure of the degree of disorder of a system is known as
- 2The pressure in the tyre of a car is four times the atmospheric pressure at $300 K$. If this tyre suddenly bursts, its new temperature will be $(\gamma = 1.4)$View Solution
- 3A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
- 4$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
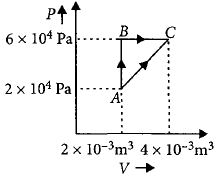
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 5Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
- 6A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 7View SolutionWhich is the correct statement
- 8View SolutionWhich of the following can be coefficient of performance of refrigerator?
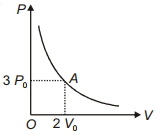
- 9The variation of pressure $P$ with volume $V$ for an ideal monatomic gas during an adiabatic process is shown in figure. At point $A$ the magnitude of rate of change of pressure with volume isView Solution
- 10The efficiency of a thermodynamic cycle $1-2-3- 1 ($see picture$)$ is $20\%$ and for another thermodynamic cycle $1 - 3-4 - 1$ efficiency is equal to $10\%$. Determine the efficiency $\eta ($ in $\%)$ of the thermodynamic cycle $1-2-3-4- 1.$The gas is assumed to be idealView Solution