Irreversible process is
Easy
The method of expansion in which a gas or liquid at pressure $P_1$ flows into a region of lower pressure $P_2$ without significant change in kinetic energy, is called the Joule-Thomson expansion. The expansion is inherently irreversible.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 2The gas law $\frac{{PV}}{T} = $ constant is true forView Solution
- 3The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 4View SolutionA gas is compressed adiabatically till its temperature is doubled. The ratio of its final volume to initial volume will be
- 5If a Carnot engine works between $127^{\circ} C$ and $527^{\circ} C$, then its efficiency is ...... $\%$View Solution
- 6View SolutionCofficient of performance of refigerator is
- 7An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
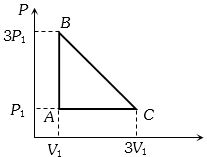
- 8A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 9Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 10A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine isView Solution
