$[\mathrm{as} W=p \Delta V \text { and } \Delta V=0, \text { so } W=0]$
$\Delta U=m C \Delta T$
$=100 \times 10^{-3} \times 4184 \times(50-30) \approx 8.4 \mathrm{kJ}$
Download our appand get started for free
Similar Questions
- 1A diatomic gas undergoes a process represented by $PV ^{1.3}=$ constant. Choose the incorrect statementView Solution
- 2A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 3Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
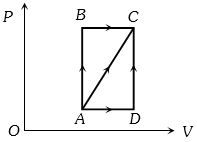
- 4A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 5A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
- 6The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 7One mole of helium is adiabatically expanded from its initial state $({P_i},{V_i},{T_i})$ to its final state $({P_f},{V_f},{T_f})$. The decrease in the internal energy associated with this expansion is equal toView Solution
- 8View SolutionEntropy of a thermodynamic system does not change when this system is used for
- 9View SolutionA cycle tyre bursts suddenly. This represents an
- 10Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
