$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following parameters does not characterize the thermodynamic state of matter
- 2In an Isobaric process, the work done by a di$-$atomic gas is $10\, J ,$ the heat given to the gas will be (in $J$)View Solution
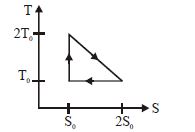
- 3View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 4View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 5A Carnot engine works first between $200^{\circ} C$ and $0^{\circ} C$ and then between $0^{\circ} C$ and $-200^{\circ} C$. The ratio of its efficiency in the two cases isView Solution
- 6One mole of an ideal gas at an initial temperature of $T\, K$ does $6\, R\, joules$ of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $\frac{5}{3}$ , the final temperature of gas will beView Solution
- 7Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
- 8View SolutionFor an ideal gas, in an isothermal process
- 9View SolutionThe slopes of isothermal and adiabatic curves are related as
- 10The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution