This cycle is one of the foundations of the second law of thermodynamics, and Carnot is often considered the father of thermodynamics. He was one of the pioneers who first determined an idealistic way of converting heat energy into work done. Carnot cycle is one of the most efficient heat engines.
Carnot cycle consists of the following four processes:
$I.$ The gas goes through an isothermal expansion at a high temperature. In this process the gas takes $q_{\text {in }}$ amount of heat from the surrounding and does $w_1$ amount of work on the surrounding.
$II.$ The gas then undergoes a reversible adiabatic expansion. Hence, the temperature of the gas comes down to a lower temperature $T_{\text {low }}$.
$III.$ Then the gas is compressed isothermally at $T_{\text {low }}$ temperature. In this process, the gas loses $q_{\text {out }}$ amount of heat, and surroundings do work on the gas.
$IV.$ Now the gas goes through a reversible adiabatic compression which makes the temperature rise up to $T_{\text {high }}$.
The following diagram shows the $P-V$ diagram of the Carnot's cycle.
Hence, Carnot's cycle consists of two isothermal and two adiabatic processes.
Download our appand get started for free
Similar Questions
- 1The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
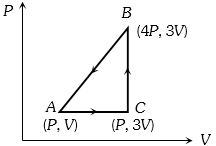
- 2A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
- 3The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
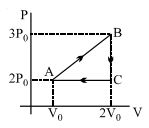
- 4View SolutionIn the above thermodynamic process, the correct statement is
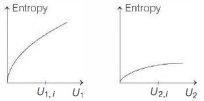
- 5Graphs below show the entropy versus energy $U$ of two systems $1$ and $2$ at constant volume. The initial energies of the systems are indicated by $U_{1, i}$ and $U_{2, i}$, respectively. Graphs are drawn to the same scale. The systems are then brought into thermal contact with each other. Assume that, at all times the combined energy of the two systems remains constant. Choose the most appropriate option indicating the energies of the two systems and the total entropy after they achieve the equilibrium.View Solution
- 6A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 7A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
- 8A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
- 9Given that $1\,g$ of water in liquid phase has volume $1\,cm^3$ and in vapour phase $1671\, cm^3$ at atmospheric pressure and the latent heat of vaporization of water is $2256\,J/g;$ the change in the internal energy in joules for $1\,g$ of water at $373\,K$ when it changes from liquid phase to vapour phase at the same temperature is ....... $J$View Solution
- 10Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
