A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, then
Easy
(b)
$d Q=\text { positive, } d U=\text { zero, } d W=\text { positive }$
$\because d Q=d U+d W$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
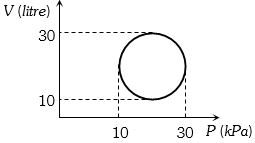
- 2If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 3A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 4A gas is compressed from a volume of $2\,m^3$ to a volume of $1\, m^3$ at a constant pressure of $100\, N/m^2$. Then it is heated at constant volume by supplying $150\, J$ of energy. As a result, the internal energy of the gasView Solution
- 5A reversible heat engine converts one-fourth of the heat input into work. When the temperature of the sink is reduced by $52\, K$, its efficiency is doubled. The temperature in Kelvin of the source will be ...... .View Solution
- 6The heat capacity of one mole an ideal is found to be $C_V=3 R(1+a R T) / 2$, where $a$ is a constant. The equation obeyed by this gas during a reversible adiabatic expansion isView Solution
- 7An ideal Carnot heat engine with an efficiency of $30\%$.It absorbs heat from a hot reservoir at $727^o C$. The temperature of the cold reservoir is .... $^oC$View Solution
- 8$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 9View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 10In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
