A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$
Medium
$\eta=25 \% \quad \eta=\frac{\mathrm{T}_{1}-\mathrm{T}_{2}}{\mathrm{T}_{1}} \times 100 \%$
$\therefore \frac{80}{\mathrm{T}_{1}} \times 100=25 \quad \Rightarrow \mathrm{T}_{1}=320 \mathrm{K}$
$\therefore \mathrm{T}_{2}=320-80=240 \mathrm{K}$
$=-33^{\circ} \mathrm{C}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
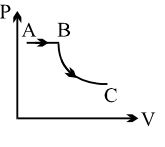
- 2View SolutionA process is shown in the diagram. Which of the following curves may represent the same process ?
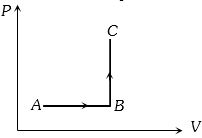
- 3The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
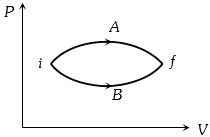
- 4In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
- 5In an adiabatic change, the pressure $P$ and temperature $T$ of a monoatomic gas are related by the relation $P \propto {T^C}$, where $C$ equalsView Solution
- 6A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 7Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
- 8A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 9A monatornic gas at a pressure $P,$ having a volume $V$ expands isothermally to a volume $2\, V$ and then adiabatically to a volume $16\, V.$ The final pressure of the gas is $(\,Take \,\gamma = 5/3)$View Solution
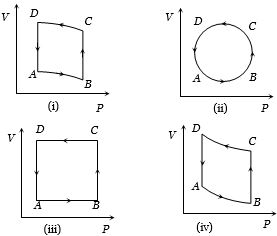
- 10In the diagrams $(i)$ to $(iv)$ of variation of volume with changing pressure is shown. A gas is taken along the path $ABCD. $ The change in internal energy of the gas will beView Solution