A monatornic gas at a pressure $P,$ having a volume $V$ expands isothermally to a volume $2\, V$ and then adiabatically to a volume $16\, V.$ The final pressure of the gas is $(\,Take \,\gamma = 5/3)$
AIPMT 2014, Medium
First, isothermal expansion
$PV = P'\left( {2V} \right);\,\,P' = \frac{P}{2}$
Then, adiabatic expansion
$P'{\left( {2V} \right)^\gamma } = {P_f}{\left( {16V} \right)^\gamma }$
$\left( {For\,adiabatic\,process,\,P{V^\gamma } = constant} \right)$
$\frac{P}{2}{\left( {2V} \right)^{5/3}} = {P_f}{\left( {16V} \right)^{5/3}}$
${P_f} = \frac{P}{2}{\left( {\frac{{2V}}{{16V}}} \right)^{5/3}} = \frac{P}{2}{\left( {\frac{1}{8}} \right)^{5/3}} = \frac{P}{2}{\left( {\frac{1}{{{2^3}}}} \right)^{5/3}}$
$ = \frac{P}{2}\left( {\frac{1}{{{2^5}}}} \right) = \frac{P}{{64}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 2One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$ 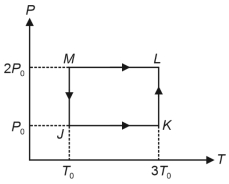
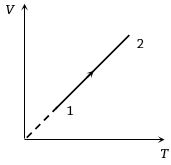
- 3Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
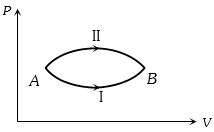
- 4A system goes from $A$ to $B$ via two processes $I$ and $II$ as shown in figure. If $\Delta {U_1}$ and $\Delta {U_2}$ are the changes in internal energies in the processes $I$ and $II$ respectively, thenView Solution
- 5View SolutionWhen heat is given to a gas in an isothermal change, the result will be
- 6An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 7A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 8A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 9View SolutionThe specific heat of a gas in an isothermal process is
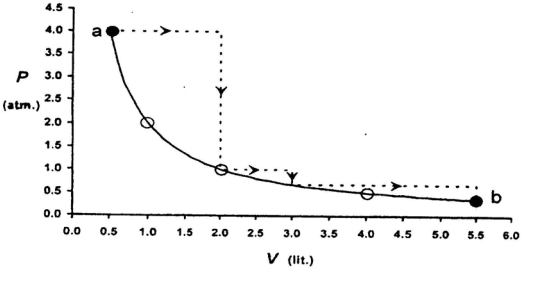
- 10One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution