A Carnot engine whose heat $\operatorname{sinks}$ at $27\,^{\circ} C$, has an efficiency of $25 \%$. By how many degrees should the temperature of the source be changed to increase the efficiency by $100 \%$ of the original efficiency $?$
JEE MAIN 2022, Diffcult
$1-\frac{300}{ T }=0$
$\frac{300}{ T }=0.75$
$1-\frac{300}{ T }=0$
$\frac{300}{ T }=0.75$
$T =400 K$
If efficiency increased by $\,100 \%$ then new efficiency $\Rightarrow n ^{\prime}=50 \%$
$1-\frac{300}{T^{\prime}}=0.5$
$T^{\prime}=600 \,K$
Increase in temp $=600-400$
$=200\, K \text { or } 200^{\circ} C$
$T =400\, K$
If efficiency increased by $100 \%\,$ then new efficiency $\Rightarrow n ^{\prime}=50 \%$
$1-\frac{300}{T^{\prime}}=0.5$
$T^{\prime}=600\,K$
Increase in temp $=600-400$
$=200 K \text { or } 200\,^{\circ} C$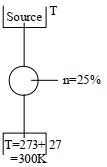
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
- 2If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
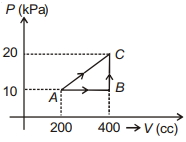
- 3The above $P-V$ diagram represents the thermodynamic cycle of an engine, operating with an ideal monatomic gas. The amount of heat, extracted from the source in a single cycle isView Solution
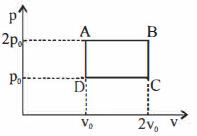
- 4A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
- 5Avessel with open mouth contains air at $60^oC$. When the vessel is heated upto temperature $T$, one fourth of the air goes out. The value of $T$ is ..... $^oC$View Solution
- 6Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

- 7An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 8The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
- 9An engine takes in $5$ moles of air at $20\,^{\circ} C$ and $1$ $atm,$ and compresses it adiabaticaly to $1 / 10^{\text {th }}$ of the original volume. Assuming air to be a diatomic ideal gas made up of rigid molecules, the change in its internal energy during this process comes out to be $X\, kJ$. The value of $X$ to the nearest integer isView Solution
- 10Air is filled in a motor tube at ${27^o}C$ and at a pressure of $8$ atmospheres. The tube suddenly bursts, then temperature of air is $[{\rm{Given}}\,\,\gamma \,{\rm{of}}\,{\rm{air}} = \,1.5]$View Solution
