A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,
KVPY 2010, Diffcult
(c)
Copper loses entropy and cools. Pond gains same amount of heat and there is some agitation in pond's water caused by plunging of copper.
Hence, pond gain more entropy than copper.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of an ideal gas is taken from a to $b$ along two paths denoted by the solid and the dashed lines as shown in the graph below. If the work done along the solid line path is $\mathrm{w}_{\mathrm{s}}$ and that along the dotted line path is $w_d$, then the integer closest to the ratio $w_d / w_5$ isView Solution
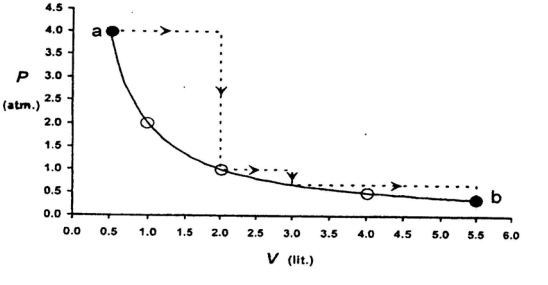
- 2View SolutionOut of the following which quantity does not depend on path
- 3A thermodynamic process is the pressure and volumes corresponding to some points in the figure are, $P_A = 3 \times 10^4 Pa$, $V_A = 2 \times 10^{-3}\, m^3$, $P_B = 8 \times 10^4 Pa$, $V_D = 5 \times 10^{-3}\,m^3$. In process $AB, 600\, J$ of heat and in process $BC, 200\, J$ of heat is added to the system. The change in the internal energy in process $AC$ would be .... $J$View Solution
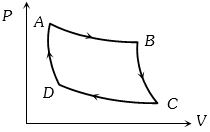
- 4The $P-V$ graph of an ideal gas cycle is shown here as below. The adiabatic process is described byView Solution
- 5The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
- 6View SolutionBy opening the door of a refrigerator placed inside a room you
- 7During an adiabatic expansion of $2\, moles$ of a gas, the change in internal energy was found $-50J.$ The work done during the process is ...... $J$View Solution
- 8Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
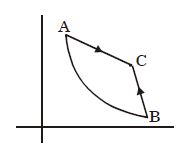
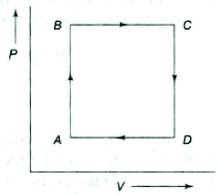
- 9View SolutionAn ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
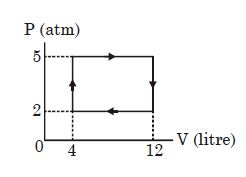
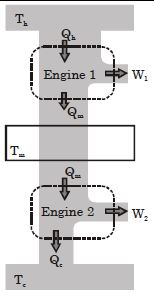
- 10Suppose that two heat engines are connected in series, such that the heat released by the first engine is used as the heat absorbed by the second engine, as shown in figure. The efficiencies of the engines are $\in_1$ and $\in_2$, respectively. The net efficiency of the combination is given by :View Solution