Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$


AIEEE 2009, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

- 2View SolutionAir in a cylinder is suddenly compressed by a piston, which is then maintained at the same position. With the passage of time
- 3Work done by a Carnot engine operating between temperatures $127^{\circ}\,C$ and $27^{\circ}\,C$ is $2\,kJ$. The amount of heat transferred to the engine by the reservoir is $........\,kJ$View Solution
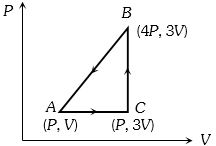
- 4A sample of ideal monoatomic gas is taken round the cycle $ABCA$ as shown in the figure. The work done during the cycle isView Solution
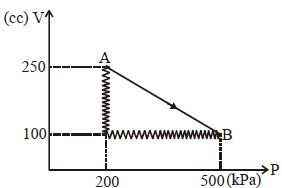
- 5A monoatomic gas is taken along path $AB$ as shown. Calculate change in internal energy of systemView Solution
- 6A Carnot's engine used first an ideal monoatomic gas then an ideal diatomic gas. If the source and sink temperature are ${411^o}C$ and ${69^o}C$ respectively and the engine extracts $1000\, J $ of heat in each cycle, then area enclosed by the $PV$ diagram is ........ $J$View Solution
- 7A piece of hot copper at $100^{\circ} C$ is plunged into a pond at $30^{\circ} C$. The copper cools down to $30^{\circ} C$, while the pond being huge stays at its initial temperature. Then,View Solution
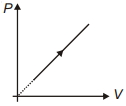
- 8View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 9An ideal gas heat engine operates in Carnot cycle between $227°C$ and $127°C.$ It absorbs $6 \times {10^4}$ cals of heat at higher temperature. Amount of heat converted to work is .........$ \times {10^4}\; cal$View Solution
- 10If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
