Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In an isolated system the entropy increases.View Solution
$Reason :$ The processes in an isolated system are adiabatic. - 2A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 3An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
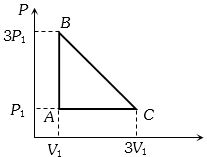
- 4Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
- 5View SolutionAn ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
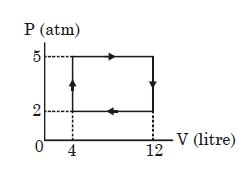
- 6View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
- 7A gas is compressed at a constant pressure of $50N/{m^2}$ from a volume of $10{m^3}$ to a volume of $4{m^3}$. Energy of $100 J$ then added to the gas by heating. Its internal energy isView Solution
- 8An ideal gas undergoes a four step cycle as shown in the $P-V$ diagram below. During this cycle, in which step heat is absorbed by the gasView Solution
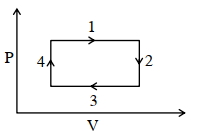
- 9View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
- 10The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will beView Solution
