Download our appand get started for free
Similar Questions
- 1A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 2A cyclic process for $1\,mole$ of an ideal gas is shown. Find work done in $AB, BC$ and $CA$ respectivelyView Solution
- 3In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 4A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given byView Solution
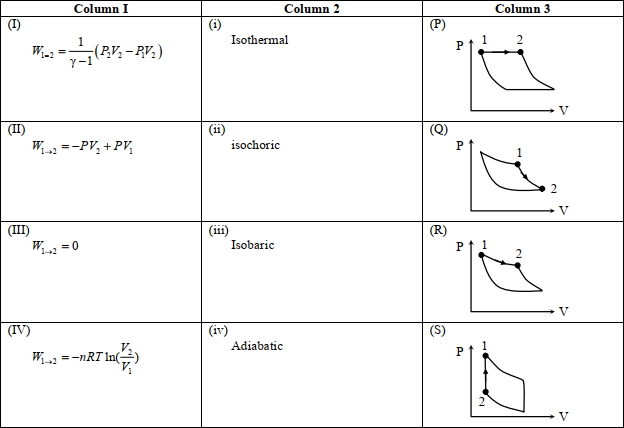
- 5An ideal gas is undergoing a cyclic thermodynamic process in different ways as shown in the corresponding $P$ $V$ diagrams in column $3$ of the table. Consider only the path from state $1$ to $2 . W$ denotes the corresponding work done on the system. The equations and plots in the table have standard notations as used in thermodynamic processes. Here $\gamma$ is the ratio of heat capacities at constant pressure and constant volume. The number of moles in the gas is $n$.View Solution
(image)
($1$) Which of the following options is the only correct representation of a process in which $\Delta U=\Delta Q-P \Delta V$ ?
$[A] (II) (iv) (R)$ $[B] (II) (iii) (P)$ $[C] (II) (iii) (S)$ $[D] (III) (iii) (P)$
($2$) Which one of the following options is the correct combination?
$[A] (III) (ii) (S)$ $[B] (II) (iv) (R)$ $[C] (II) (iv) (P)$ $[D] (IV) (ii) (S)$
($3$) Which one of the following options correctly represents a thermodynamic process that is used as a correction in the determination of the speed of sound in an ideal gas?
$[A] (III) (iv) (R)$ $[B] (I) (ii)$ $(\mathrm{Q})$ $[C] (IV) (ii) (R)$ $[D] (I) (iv) (Q)$
- 6One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 7In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 8Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
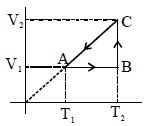
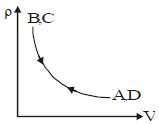
- 9Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
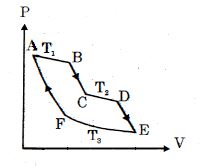
- 10One mole of ideal gas taken through a cycle process with alternate isothermal and adiabatic curves. In $P-V$ diagram $AB, CD, EF$ are isothermal curves at the absolute temperature $T_1, T_2$ and $T_3$ respectively and $BC, DE$ and $FA$ are adiabatic curves respectively. If $\frac{{{V_B}}}{{{V_A}}} = 2,\,\frac{{{V_D}}}{{{V_C}}} = 2$ then for cycle is shown in figure four statements are being made given below. (Figure is not drawn on scale)View Solution
Statement $1$ : Ratio of volumes $\frac{{{V_E}}}{{{V_F}}} = 4$
Statement $2$ : Magnitude of work done in isothermal compression $EF$ is $2RT_3\ ln\ (2)$
Statement $3$ : Ratio of heat supplied to gas in the process $AB$ to heat rejected by gas in process $EF$ is $\frac{{{T_1}}}{{2{T_3}}}$
Statement $4$ : Net work done by gas in the cycle $ABCDEFA$ is $(T_1 + T_2 - 2T_3) R\ ln\ (2)$
Find the number of correct statement $(s)$ given for the cyclic process followed by gas