A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given by
IIT 2000,JEE MAIN 2021, Diffcult
(d) ${T_1}{V_1}^{\gamma - 1} = {T_2}{V_2}^{\gamma - 1}$$ \Rightarrow \frac{{{T_1}}}{{{T_2}}} = {\left( {\frac{{{V_2}}}{{{V_1}}}} \right)^{\,\gamma - 1}}$$ = {\left( {\frac{{{L_2}A}}{{{L_1}A}}} \right)^{\,\frac{5}{3} - 1}} = {\left( {\frac{{{L_2}}}{{{L_1}}}} \right)^{\,\frac{2}{3}}}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$View Solution
- 2When an ideal gas $(\gamma = 5/3$) is heated under constant pressure, then what percentage of given heat energy will be utilised in doing external workView Solution
- 3View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
- 4One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$View Solution
- 5One mole of an ideal monoatomic gas is heated at a constant pressure of one atmosphere from ${0^o}C$ to ${100^o}C$. Then the change in the internal energy isView Solution
- 6An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 7If ${C_V} = 4.96cal/mole\, K$, then increase in internalenergy when temperature of $2$ moles of this gas is increased from $340 K$ to $342 K$ ....... $cal$View Solution
- 8View SolutionIn Carnot engine the work done by working substance is equivalent to
- 9When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
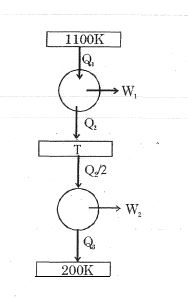
- 10Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution