A Carnot’s engine is made to work between $200°C$ and $0°C$ first and then between $0°C$ and $-200°C.$ The ratio of efficiencies of the engine in the two cases is
Medium
(b) In first case ${\eta _1} = 1 - \frac{{{T_2}}}{{{T_1}}} = 1 - \frac{{(273 + 0)}}{{(273 + 200)}} = \frac{{200}}{{473}}$
In second case ${\eta _2} = 1 - \frac{{(273 - 200)}}{{(273 + 0)}} = \frac{{200}}{{273}}$
==> $\frac{{{\eta _1}}}{{{\eta _2}}} = \frac{1}{{\left( {\frac{{473}}{{273}}} \right)}} = 1:1.73$
In second case ${\eta _2} = 1 - \frac{{(273 - 200)}}{{(273 + 0)}} = \frac{{200}}{{273}}$
==> $\frac{{{\eta _1}}}{{{\eta _2}}} = \frac{1}{{\left( {\frac{{473}}{{273}}} \right)}} = 1:1.73$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
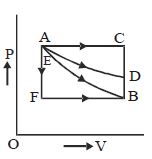
- 1An ideal monoatomic gas is taken round the cycle $12341$ shown in the figure ( $p\,-\,V$ diagram). The work done in one cycle is ?View Solution
- 2Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 3An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 4$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 5For a thermodynamic process $\delta Q = -50$ $calorie$ and $W = -20$ $calorie$ . If the initial internal energy is $-30$ $calorie$ then final internal energy will be ....... $calorie$View Solution
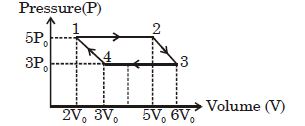
- 6Which of the following is an equivalent cyclic process corresponding to the thermodynamic cyclic given in the figure? where, $1 \rightarrow 2$ is adiabatic.View Solution
(Graphs are schematic and are not to scale)
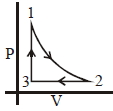
- 7In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 8$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 9If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 10An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution