A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?
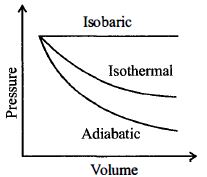

JEE MAIN 2013, Medium
Work done $=$ Area bounded by $PV$ graph and volume axis
Among the three process, bounded area by $PV$ graph and volume axis is greatest for isobaric process, hence work done is greatest for isobaric process
Among the three process, bounded area by $PV$ graph and volume axis is greatest for isobaric process, hence work done is greatest for isobaric process
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
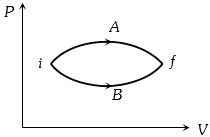
- 1In the figure given two processes $A$ and $B$ are shown by which a thermo-dynamical system goes from initial to final state $F.$ If $\Delta {Q_A}$ and $\Delta {Q_B}$ are respectively the heats supplied to the systems thenView Solution
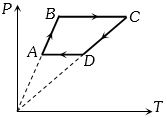
- 2Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution
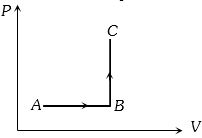
- 3The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
- 4$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 5Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 6During an adiabatic process, the pressure of a gas is found to be proportional to the cube of its temperature. The ratio of $\frac{{{C_P}}}{{{C_V}}}$ for the gas isView Solution
- 7Two carnot engines $A$ and $B$ operate in series such that engine $A$ absorbs heat at $T_{1}$ and rejects heat to a sink at temperature $T$. Engine $B$ absorbs half of the heat rejected by engine $A$ and rejects heat to the sink at ${T}_{3}$. When workdone in both the cases is equal, the value of ${T}$ isView Solution
- 8View SolutionIn the following figures heat is absorbed by the gas
- 9$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 10View SolutionNeon gas of a given mass expands isothermally to double volume. What should be the further fractional decrease in pressure, so that the gas when adiabatically compressed from that state, reaches the original state?
