A Carnot engine whose low temperature reservoir is at $7\,°C$ has an efficiency of $50\%$. It is desired to increase the efficiency to $70\%$. By how many degrees should the temperature of the high temperature reservoir be increased ....... $K$
Medium
(d) Initially $\eta = \frac{{{T_1} - {T_2}}}{{{T_1}}}$==> $0.5 = \frac{{{T_1} - (273 + 7)}}{{{T_1}}}$
==> $\frac{1}{2} = \frac{{{T_1} - 280}}{{{T_1}}}$==> ${T_1} = 560K$
Finally ${\eta _1}' = \frac{{{T_1}'\, - {T_2}}}{{{T_1}^\prime }}$==>$0.7 = \frac{{{T_1}'\, - \,(273 + 7)}}==>{{{T_1}^\prime }}$==>${T_1}' = 933K$
$\therefore$ increase in temperature $ = 933 - 560 = 373K \approx 380K$
==> $\frac{1}{2} = \frac{{{T_1} - 280}}{{{T_1}}}$==> ${T_1} = 560K$
Finally ${\eta _1}' = \frac{{{T_1}'\, - {T_2}}}{{{T_1}^\prime }}$==>$0.7 = \frac{{{T_1}'\, - \,(273 + 7)}}==>{{{T_1}^\prime }}$==>${T_1}' = 933K$
$\therefore$ increase in temperature $ = 933 - 560 = 373K \approx 380K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 2A Carnot engine takes $3 \times {10^6}\,cal$. of heat from a reservoir at $627°C$, and gives it to a sink at $27°C.$ The work done by the engine isView Solution
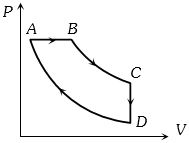
- 3A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 4A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 5A solid body of constant heat capacity $1\ J/^o C$ is being heated by keeping it in contact with reservoirs in two ways :View Solution
$(i)$ Sequentially keeping in contact with $2$ reservoirs such that each reservoir supplies same amount of heat.
$(ii)$ Sequentially keeping in contact with $8$ reservoirs such that each reservoir supplies same amount of heat.
In both the cases body is brought from initial temperature $100^o C$ to final temperature $200^o C$. Entropy change of the body in the two cases respectively is :
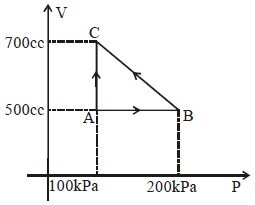
- 6The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
- 7View SolutionIn the following figures heat is absorbed by the gas
- 8The efficiency of carnot engine is $50\%$ and temperature of sink is $500\;K$. If temperature of source is kept constant and its efficiency raised to $60\%$, then the required temperature of the sink will beView Solution
- 9$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours. - 10A cyclic process $ABCD$ is shown in the figure $P-V$ diagram. Which of the following curves represent the same processView Solution