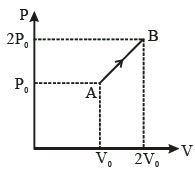
$\Delta \mathrm{Q}=\Delta \mathrm{W}+\Delta \mathrm{V}$
$\Delta \mathrm{W}=\frac{1}{2} \times\left(2 \mathrm{P}_{0}+\mathrm{P}_{0}\right) \times \mathrm{V}_{0}=\frac{3}{2} \mathrm{P}_{0} \mathrm{V}_{0}$
$\frac{\mathrm{PV}}{\mathrm{T}}=\mathrm{const}=\mathrm{nR}$
$\frac{P_{0} V_{0}}{T_{0}}=\frac{2 P_{0} 2 V_{0}}{T^{\prime}}$
$\mathrm{T}^{\prime}=4 \mathrm{T}_{\mathrm{o}}$
change in temperature $=3 \mathrm{T}_{0}$
$\Delta \mathrm{U}=\mathrm{n} \times \frac{3}{2} \mathrm{R} \times 3 \mathrm{T}_{0}=\frac{9}{2}\left(\mathrm{nRT}_{0}\right)=\frac{9}{2} \mathrm{P}_{0} \mathrm{V}_{0}$
$\Delta \mathrm{Q}=6 \mathrm{P}_{0} \mathrm{V}_{0}$
Download our appand get started for free
Similar Questions
- 1A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
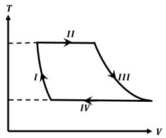
- 2One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
- 3$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 4A Carnot engine has an efficiency of $50 \%$ when its source is at a temperature $327^{\circ}\,C$. The temperature of the sink is $.........^{\circ} C$View Solution
- 5One gm mol of a diatomic gas $(\gamma = 1.4)$ is compressed adiabatically so that its temperature rises from ${27^o}C$ to ${127^o}C$. The work done will beView Solution
- 6View SolutionA measure of the degree of disorder of a system is known as
- 7View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
- 8For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
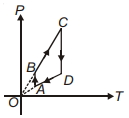
- 9A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
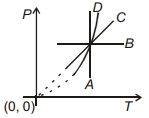
- 10Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution