A closed vessel contains $0.1$ mole of a monoatomic ideal gas at $200\, K$. If $0.05$ mole of the same gas at $400\, K$ is added to it, the final equilibrium temperature (in $K$ ) of the gas in the vessel will be closed to
JEE MAIN 2020, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 2$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):View Solution
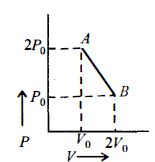
- 3$n\, moles$ of an ideal gas undergo a process $A \to B$ as shown in the figure. Maximum temperature of the gas during the process isView Solution
- 4When $1\, kg$ of ice at $0^o C$ melts to water at $0^o C,$ the resulting change in its entropy, taking latent heat of ice to be $80\, cal/gm,$ is ...... $cal/K$View Solution
- 5View Solution“Heat cannot by itself flow from a body at lower temperature to a body at higher temperature” is a statement or consequence of
- 6Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 7The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$View Solution
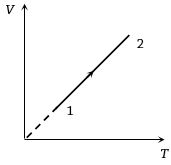
- 8Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 9View SolutionOut of the following which quantity does not depend on path
- 10In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
