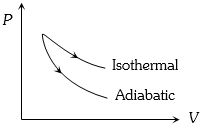
Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$1\,g$ of a liquid is converted to vapour at $3 \times 10^5\,Pa$ pressure. If $10 \%$ of the heat supplied is used for increasing the volume by $1600\,cm ^3$ during this phase change, then the increase in internal energy in the process will be $............\,J$View Solution
- 2$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
$Reason :$ Dissipative effects cannot be eliminated. - 3Given below are two statementView Solution
Statement $-I$ : What $\mu$ amount of an ideal gas undergoes adiabatic change from state $\left( P _{1}, V _{1}, T _{1}\right)$ to state $\left( P _{2}, V _{2}, T _{2}\right)$, the work done is $W =\frac{1 R \left( T _{2}- T _{1}\right)}{1-\gamma}$, where $\gamma=\frac{ C _{ P }}{ C _{ V }}$ and $R =$ universal gas constant,
Statement $-II$ : In the above case. when work is done on the gas. the temperature of the gas would rise.
Choose the correct answer from the options given below
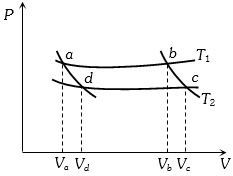
- 4In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
- 5A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
- 6If minimum possible work is done by a refrigerator in converting $100\; grams$ of water at $0^{\circ} C$ to ice, how much heat (in calories) is released to the surrounding at temperature $27^{\circ} C$ (Latent heat of ice $=80 Cal / gram$ ) to the nearest integer?View Solution
- 7An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 8$Assertion :$ In isothermal process whole of the heat supplied to the body is converted into internal energy.View Solution
$Reason :$ According to the first law of thermodynamics : $\Delta Q = \Delta U + p\Delta V$ - 9Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 10A monatornic gas at a pressure $P,$ having a volume $V$ expands isothermally to a volume $2\, V$ and then adiabatically to a volume $16\, V.$ The final pressure of the gas is $(\,Take \,\gamma = 5/3)$View Solution