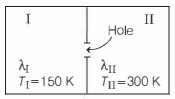
A container is divided into two equal parts $I$ and $II$ by a partition with a small hole of diameter $d$. The two partitions are filled with same ideal gas, but held at temperatures $T_{ I }=150 \,K$ and $T_{ II }=300 \,K$ by connecting to heat reservoirs. Let $\lambda_{1}$ and $\lambda_{1 I}$ be the mean free paths of the gas particles in the two parts, such that $d >> \lambda_{ I }$ and $d >> \lambda_{ II }$. Then, the $\lambda_{ I } / \lambda_{ II }$ is close to
KVPY 2016, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$5$ moles of oxygen is heated at constant volume from $10°C$ to $20°C.$ The change in the internal energy of the gas is (the gram molecular specific heat of oxygen at constant pressure...... $cal$ ${C_p} = 8$ $cal/mole \,°C$ and $R = 8.3 cal/mole\, °C)$View Solution
- 2The average speed $v$ and $r.m.s.$ speed $\bar v$ of the molecules are related asView Solution
- 3View SolutionAt the same temperature and pressure and volume of two gases, which of the following quantities is constant
- 4View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 5A given mass of a gas is allowed to expand freely until its volume becomes double. If ${C_b}$ and ${C_a}$ are the velocities of sound in this gas before and after expansion respectively, then ${C_a}$ is equal toView Solution
- 6The total internal energy of two mole monoatomic ideal gas at temperature $T=300\,K$ will be ...........$J$.View Solution
$\text { (Given } R =8.31\,J / mol.K )$
- 7If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
- 8At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 9View SolutionInternal energy of a non-ideal gas depends on ..........
- 10For a gas, the $r.m.s.$ speed at $800\, K$ isView Solution