The total internal energy of two mole monoatomic ideal gas at temperature $T=300\,K$ will be ...........$J$.
$\text { (Given } R =8.31\,J / mol.K )$
JEE MAIN 2022, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Molar specific heat at constant volume is ${C_v}$ for a monoatomic gas isView Solution
- 2The energy (in $eV$) possessed by a neon atom at $27^{\circ} C$ is ..............View Solution
- 3The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
- 4To what temperature should the hydrogen at room temperature $(27°C)$ be heated at constant pressure so that the $R.M.S.$ velocity of its molecules becomes double of its previous value ....... $^oC$View Solution
- 5A narrow glass tube, $80 \,cm$ long and opens at both ends, is half immersed in mercury, now the top of the tube is closed and is taken out of mercury. A column of mercury $20 \,cm$ long remains in the tube. Find atmospheric pressureView Solution
- 6The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 7The value of densities of two diatomic gases at constant temperature and pressure are ${d_1}$ and ${d_2}$, then the ratio of speed of sound in these gases will beView Solution
- 8Two vessels of the same volume contain the same gas at same temperature. If the pressure in the vessels be in the ratio of $1 : 2$, thenView Solution
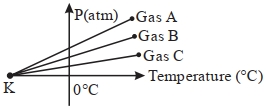
- 9For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$
- 10$c_P$ and $c_V$ are specific heats at constant pressure and constant volume respectively. It is observed thatView Solution
$c_P - c_V = a$ for hydrogen gas
$c_P - c_V = b$ for nitrogen gas
The correct relation between $a$ and $b$ is
