A container $X$ has volume double that of contianer $Y$ and both are connected by a thin tube. Both contains same ideal gas. The temperature of $X$ is $200\,\,K$ and that of $Y$ is $400\,\,K$. If mass of gas in $X$ is $m$ then in $Y$ it will be:
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container of fixed volume has a mixture of one mole of hydrogen and one mole of helium in equilibrium at temperature $T$. Assuming the gases are ideal, the correct statement$(s)$ is(are)View Solution
$(A)$ The average energy per mole of the gas mixture is $2RT$.
$(B)$ The ratio of speed of sound in the gas mixture to that in helium gas is $\sqrt{6 / 5}$.
$(C)$ The ratio of the rms speed of helium atoms to that of hydrogen molecules is $1 / 2$.
$(D)$ The ratio of the rms speed of helium atoms to that of hydrogen molecules is $1 / \sqrt{2}$.
- 2A $25\times10^{-3}\, m^3$ volume cylinder is filled with $1\, mol$ of $O_2$ gas at room temperature $(300\, K)$. The molecular diameter of $O_2$, and its root mean square speed, are found to be $0.3\, nm$ and $200\, m/s$, respectively. What is the average collision rate (per second) for an $O_2$ molecule?View Solution
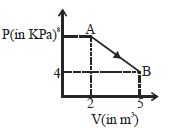
- 3A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 4Read the given statements and decide which is/are correct on the basis of kinetic theory of gasesView Solution
$(I)$ Energy of one molecule at absolute temperature is zero
$(II)$ $r .m.s.$ speeds of different gases are same at same temperature
$(III)$ For one gram of all ideal gas kinetic energy is same at same temperature
$(IV)$ For one mole of all ideal gases mean kinetic energy is same at same temperature
- 5The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 6At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 7$Assertion :$ Air pressure in a car tyre increases during driving.View Solution
$Reason :$ Absolute zero temperature is not zero energy temperature. - 8The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 9The average degree of freedom per molecule of a gas is $6.$ The gas performs $25 \,J$ work, while expanding at constant pressure. The heat absorbed by the gas is ...... $J$View Solution
- 10If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
