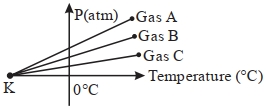
A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
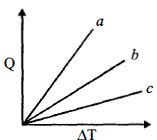
- 1Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 2One gram mole of an ideal gas $A$ with the ratio of constant pressure and constant volume specific heats $\gamma_{A}=5 / 3$ is mixed with $n$ gram moles of another ideal gas $B$ with $\gamma_{B}=7 / 5$. If the $\gamma$ for the mixture is $19 / 13$, then what will be the value of $n$ ?View Solution
- 3$C{O_2}(O - C - O)$ is a triatomic gas. Mean kinetic energy of one gram gas will be (If $N-$Avogadro's number, $k-$Boltzmann's constant and molecular weight of $C{O_2} = 44$)View Solution
- 4The number of gas molecules striking per second per square metre of the top surface of a table placed in a room at $20^{\circ} C$ and 1 atmospheric pressure is of the order of $\left(k_{B}=1.4 \times 10^{-23} \,JK ^{-1}\right.$ and the average mass of an air molecule is $5 \times 10^{-27} \,kg$ )View Solution
- 5The gas in vessel is subjected to a pressure of $20$ atmosphere at a temperature $27°C.$ The pressure of the gas in a vessel after one half of the gas is released from the vessel and the temperature of the remainder is raised by $50°C$ is ....... $atm$View Solution
- 6If the root mean square velocity of the molecules of hydrogen at $NTP$ is $1.84\, km/s$. Calculate the root mean square velocity of oxygen molecule at $NTP$, molecular weight of hydrogen and oxygen are $2$ and $32$ respectively ....... $km/sec$View Solution
- 7Molecules of an ideal gas are known to have three translational degrees of freedom and two rotational degrees of freedom. The gas is maintained at a temperature of $T$. The total internal energy, $U$ of a mole of this gas, and the value of $\gamma\left(=\frac{ C _{ P }}{ C _{ v }}\right)$ given, respectively, byView Solution
- 8Using equipartition of energy, the specific heat (in $J\, kg^{-1}\, K^{-1}$ ) of aluminium at room temperature can be estimated to be ( atomic weight of aluminium $= 27$)View Solution
- 9Ideal monoatomic gas is taken through a process $dQ = 2dU.$ The molar heat capacity for the processView Solution
- 10For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$