A cylinder of $5$ litre capacity, filled with air at $N.T.P.$ is connected with another evacuated cylinder of $30$ litres of capacity. The resultant air pressure in both the cylinders will be ...... $cm$ of $Hg$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
- 2The total kinetic energy of $1$ mole of oxygen at $27^{\circ} \mathrm{C}$ is :View Solution
[Use universal gas constant $(R)=8.31 \mathrm{~J} / \mathrm{mole} \mathrm{K}$ ]
- 3A box containing $N$ molecules of a perfect gas at temperature ${T_1}$ and pressure ${P_1}$. The number of molecules in the box is doubled keeping the total kinetic energy of the gas same as before. If the new pressure is ${P_2}$ and temperature ${T_2}$, thenView Solution
- 4For three low density gases $A, B, C$ pressure versus temperature graphs are plotted while keeping them at constant volume, as shown in the figure.View Solution
The temperature corresponding to the point ' $K$ ' is $...........\,{}^{\circ}\,C$
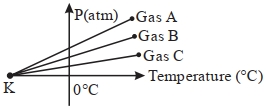
- 5We write the relation for Boyle's law in the form $PV = C$ when the temperature remains constant. In this relation, the magnitude of $C$ depends uponView Solution
- 6A gas at absolute temperature $300\,K$ has pressure $= 4 \times 10^{-10}\,N /m^2$ . Boltzmann constant, $k = 1.38 \times 10^{-23}\,J / K$ . The number of molecules per $cm^3$ is of the order ofView Solution
- 7View SolutionAt absolute zero temperature, pressure of a gas will be
- 8The root mean square speed of molecules of a given mass of a gas at $27^{\circ} C$ and $1$ atmosphere pressure is $200\, ms ^{-1}$. The root mean square speed of molecules of the gas at $127^{\circ} C$ and $2$ atmosphere pressure is $\frac{ x }{\sqrt{3}}\, ms ^{-1} .$ The value of $x$ will be ......$ms ^{-1} .$View Solution
- 9When the temperature of a gas is raised from $30^o C$ to $90^o C$ , the percentage increase in the $r.m.s.$ velocity of the molecules will beView Solution
- 10Two gases are at absolute temperatures $300 K $ and $350 K$ respectively. Ratio of average kinetic energy of their molecules isView Solution
