The isothermal bulk modulus of a perfect gas at normal pressure is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
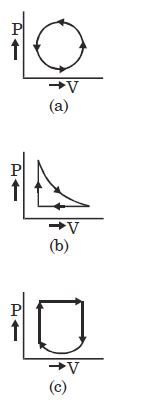
- 1View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
- 2View SolutionFirst law of thermodynamics is a special case of
- 3In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 4A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
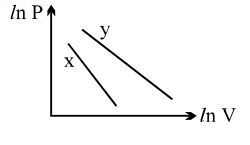
- 5For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
- 6The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
- 7$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
$Reason :$ Dissipative effects cannot be eliminated. - 8For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
- 9If one mole of an ideal gas goes through the process $A \rightarrow B$ and $B \rightarrow C .$ Given that $T _{ A }=400\, K ,$ and $T _{ C }=400 \,K .$ If $\frac{ P _{ B }}{ P _{ A }}=\frac{1}{5},$ then find the heat supplied to the gas (in $J$)View Solution
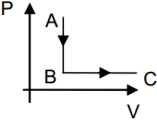
- 10View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure