One mole of an ideal gas expands at a constant temperature of $300 \,K$ from an initial volume of $10\, litres$ to a final volume of $20\, litres$. The work done in expanding the gas is ...... $J.$ $(R = 8.31 J/mole-K)$
Medium
(b) ${W_{iso}} = \mu RT{\log _e}\frac{{{V_2}}}{{{V_1}}} = 1 \times 8.31 \times 300{\log _e}\frac{{20}}{{10}} = 1728J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1During the melting of a slab of ice at $273\, K$ at atmospheric pressureView Solution
- 2View SolutionTwo gases are said to be in thermal equilibrium when they have same
- 3View SolutionWhich relation is correct for isometric process
- 4The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 5A Container having $1\ mole$ of a gas at a temperature $27\ ^oC$ has a movable piston which maintains at constant pressure in container of $1\ atm.$ The gas is compressed until temperature becomes $127^oC.$ The work done is ........ $J$ $(C_p\ for\ gas\ is\ 7.03\ cal/mol-K)$View Solution
- 6View SolutionIn thermodynamics, heat and work are
- 7The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
- 8View SolutionFirst law thermodynamics states that
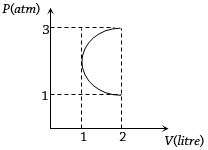
- 9In the $P-V$ diagram shown in figure $ABC$ is a semicircle. The work done in the process $ABC$ isView Solution
- 10An ideal gas heat engine operates in Carnot cycle between $227°C$ and $127°C.$ It absorbs $6 \times {10^4}$ cals of heat at higher temperature. Amount of heat converted to work is .........$ \times {10^4}\; cal$View Solution
