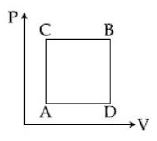
A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$

JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
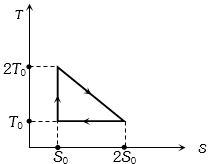
- 2View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 3'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
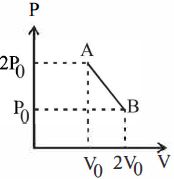
- 4A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process isView Solution
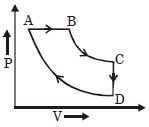
- 5An ideal refrigerator has a freezer at a temperature of $ - 13^\circ C.$ The coefficient of performance of the engine is $5.$ The temperature of the air (to which heat is rejected) will beView Solution
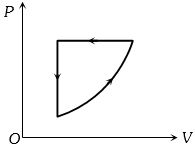
- 6View SolutionIn the above thermodynamic process, the correct statement is
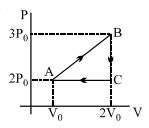
- 7View SolutionWhich statement is incorrect?
- 8$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
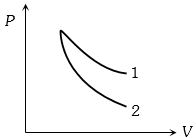
- 9For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
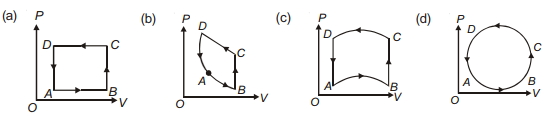
- 10In following figures $(a)$ to $(d)$, variation of volume by change of pressure is shown in figure. The gas is taken along the path $A B C D A$. Change in internal energy of the gas will be .......View Solution