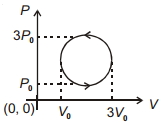
For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correct

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
- 2$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 3In an adiabatic process $90J$ of work is done on the gas. The change in internal energy of the gas is ....... $J$View Solution
- 4A Carnot engine with efficiency $50\,\%$ takes heat from a source at $600\,K$. In order to increase the efficiency to $70\,\%$, keeping the temperature of sink same, the new temperature of the source will be $.........\,K$View Solution
- 5If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
- 6A monoatomic gas is taken through a process $TP^{-1/3} =$ constant. If heat is given to the gasView Solution
- 7Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
- 8A real gas within a closed chamber at $27^{\circ} \mathrm{C}$ undergoes the cyclic process as shown in figure. The gas obeys $P V^3=\mathrm{RT}$ equation for the path $A$ to $B$. The net work done in the complete cycle is (assuming $R=8 \mathrm{~J} / \mathrm{molK}$ ):View Solution
- 9Two Carnot engines $A$ and $B$ are operated in succession. The first one, $A$ receives heat from a source at $T_1 = 800\, K$ and rejects to sink at $T_2K$. The second engine $B$ receives heat rejected by the first engine and rejects to another sink at $T_3 = 300\, K$. If the work outputs of two engines are equal, then the value of $T_2$ is ...... $K$View Solution
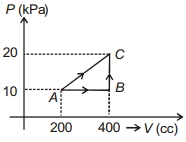
- 10View SolutionWork done in the cyclic process shown in figure is ...........