An electron tube was sealed off during manufacture at a pressure of $1.2 \times {10^{ - 7}}$ $mm$ of mercury at $27°C.$ Its volume is $100\, cm^3$. The number of molecules that remain in the tube is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe molar specific heat at constant pressure for a monoatomic gas is
- 2The root mean square speed of the molecules of a diatomic gas is $v$. When the temperature is doubled, the molecules dissociate into two atoms. The new root mean square speed of the atom isView Solution
- 3${O_2}$ gas is filled in a vessel. If pressure is doubled, temperature becomes four times. How many times its density will becomeView Solution
- 4This question has Statement $- 1$ and Statement $-2$. Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1$ : The internal energy of a perfect gas is entirely kinetic and depends only on absolute temperature of the gas and not on its pressure or volume.
Statement $2$ : A perfect gas is heated keeping pressure constant and later at constant volume. For the same amount of heat the temperature of the gas at constant pressure is lower than that at constant volume. - 5The number of molecules in a gas at pressure $1.64 \times {10^{ - 3}}$atmospheres and temperature $200\, K$ having the volume $1 \,cc$ areView Solution
- 6In Vander Waal's equation $\left[ {P + \frac{a}{{{V^2}}}} \right]\,(V - b) = RT,$ the dimensions of $a$ areView Solution
- 7At which temperature the r.m.s. velocity of a hydrogen molecule equal to that of an oxygen molecule at $47^{\circ} \mathrm{C}$ ?View Solution
- 8What is the velocity of wave in monoatomic gas having pressure $1$ kilopascal and density $2.6\,kg/{m^3}$View Solution
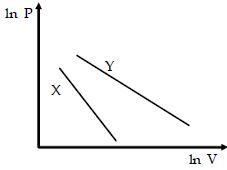
- 9The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
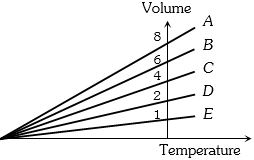
- 10For two different gases $X$ and $Y,$ having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{v_1}$ and $C_{v_2}$ respectively, for adiabatic process , the $\ln P$ versus $\ln V$ graph is plotted as shown :-View Solution