The gas in vessel is subjected to a pressure of $20$ atmosphere at a temperature $27°C.$ The pressure of the gas in a vessel after one half of the gas is released from the vessel and the temperature of the remainder is raised by $50°C$ is ....... $atm$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In Maxwell's speed distribution curve, for $N_2$ gas, the average of $\mid$ relative velocity (in $m/s$) $\mid$ between two molecules at $300 \,K$ will beView Solution
- 2View SolutionAn ideal gas is filled in a vessel, then
- 3For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
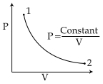
- 4For a certain process, pressure of diatomic gas varies according to the relation $P = aV^2$, where $a$ is constant. What is the molar heat capacity of the gas for this process?View Solution
- 5A diatomic gas follows equation $PV^m =$ constant, during a process. What should be the value of $m$ such that its molar heat capacity during process $= R$View Solution
- 6A diatomic gas of molecular mass $40 \,g / mol$ is filled in a rigid container at temperature $30^{\circ} C$. It is moving with velocity $200 \,m / s$. If it is suddenly stopped, the rise in the temperature of the gas is .........View Solution
- 7A gas obeying the equation of state $p V=R T$ undergoes a hypothetical reversible process described by the equation, $p V^{5 / 3} \exp \left(-\frac{p V}{E_{0}}\right)=C_{1}$, where $C_{1}$ and $E_{0}$ are dimensioned constants. Then, for this process, the thermal compressibility at high temperatureView Solution
- 8$Assertion :$ The root mean square and most probable speeds of the molecules in a gas are the same.View Solution
$Reason :$ The Maxwell distribution for the speed of molecules in a gas in symmetrical. - 9At $10^o C$ the value of the density of a fixed mass of an ideal gas divided by it pressure is $x.$ At $110^o C$ this ratio isView Solution
- 10$1$ mole of gas occupies a volume of $100\, ml$ at $50\, mm$ pressure. What is the volume occupied by two moles of gas at $100\, mm$ pressure and at same temperature ..... $ml$View Solution
