The number density of molecules of a gas depends on their distance $r$ from the origin as, $n\left( r \right) = {n_0}{e^{ - \alpha {r^4}}}$. Then the total number of molecules is proportional to
JEE MAIN 2019, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A sample of an ideal gas occupies a volume $V$ at a pressure $P$ and absolute temperature $T,$ the mass of each molecule is $m.$ The expression for the density of gas is ($k =$ Boltzmann’s constant)View Solution
- 2At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
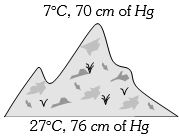
- 3At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 4At $0°C $ the density of a fixed mass of a gas divided by pressure is $x.$ At $100°C,$ the ratio will beView Solution
- 5View SolutionWe sit in the room with windows open. Then,
- 6The root mean square velocity of a gas molecule of mass $m$ at a given temperature is proportional toView Solution
- 7One mole of ideal monoatomic gas $(\gamma = 5/3)$ is mixed with one mole of diatomic gas $(\gamma = 7/5).$ What is $\gamma $ for the mixture$?$ $\gamma $ denotes the ratio of specific heat at constant pressure, to that at constant volumeView Solution
- 8The r.m.s. speed of a group of $7$ gas molecules having speeds $(6, 4, 2, 0, -2, -4, -6)\, m/s$ is ...... $m/s$View Solution
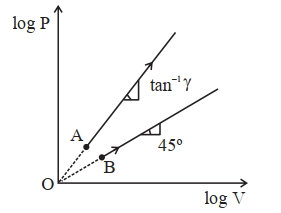
- 9Two thermodynamical process are shown in the figure. The molar heat capacity for process $A$ and $B$ are $C_A$ and $C_B$. The molar heat capacity at constant pressure and constant volume are represented by $C_P$ and $C_V$, respectively. Choose the correct statement.View Solution
- 10A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
