A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$
Easy
(b) $P{V^\gamma } = $constant ==> $\frac{{{P_2}}}{{{P_1}}} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^\gamma } = {\left( {\frac{{{V_1}}}{{{V_1}/4}}} \right)^\gamma } = {4^\gamma }$
==> ${P_2} = {4^\gamma }P$
As $\gamma$ is always greater than one so ${4^\gamma } > 4$ ==> ${P_2} > 4P$
==> ${P_2} = {4^\gamma }P$
As $\gamma$ is always greater than one so ${4^\gamma } > 4$ ==> ${P_2} > 4P$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 2In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 3A refrigerator consumes an average $35\, {W}$ power to operate between temperature $-10^{\circ} {C}$ to $25^{\circ} {C}$. If there is no loss of energy then how much average heat per second does it transfer? (in ${J} / {s}$)View Solution
- 4A sample of an ideal gas is taken through a cycle a shown in figure. It absorbs $50J$ of energy during the process $AB$, no heat during $BC$, rejects $70J$ during $CA.$ $40J$ of work is done on the gas during $BC$. Internal energy of gas at $A$ is $1500J$, the internal energy at $C$ would be ........ $J$View Solution
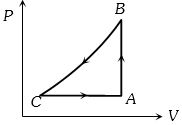
- 5A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
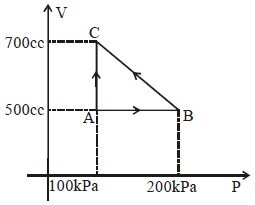
- 6$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
- 7The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 8View SolutionThe maximum possible efficiency of a heat engine is ...........
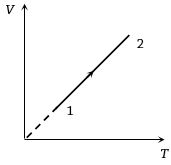
- 9Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 10A frictionless heat engine can be $100 \%$ efficient only if its exhaust temperature is ............View Solution
