A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found that
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A container of volume $1{m^3}$is divided into two equal compartments by a partition. One of these compartments contains an ideal gas at $300 K$. The other compartment is vacuum. The whole system is thermally isolated from its surroundings. The partition is removed and the gas expands to occupy the whole volume of the container. Its temperature now would be ..... $K$View Solution
- 2View SolutionA process can be reversible if
- 3The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 4View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
- 5An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
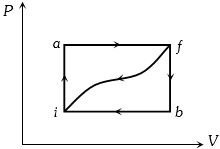
- 6When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 7Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 8For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 9An ideal gas is taken through the cycle $A \to B \to C \to A$ , as shown in the figure. If the net heat supplied to the gas in the cycle is $5\ J$, the work done by the gas in the process $C \to A$ is .... $J$View Solution
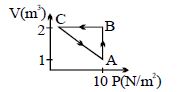
- 10A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution