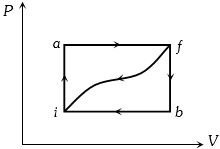
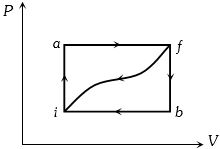
When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$

Medium
(d) $\Delta U$, remains same for both path
For path $iaf : \Delta U = \Delta Q - \Delta W = 50 - 20 = 30J$.
For path $fi : \Delta U = - \,30\,J\,{\rm{and\, }}\Delta W = - 13J$
==> $\Delta Q = - \,30 - 13 = - \,43\;J$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
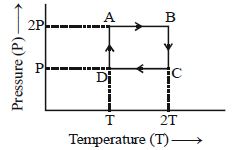
- 1One mole of an ideal gas having initial volume $V$, pressure $2P$ and temperature $T$ undergoes a cyclic process $ABCDA$ as shown below : The net work done in the complete cycle isView Solution
- 2When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 3Consider a spherical shell of radius $R$ at temperature $T$. The black body radiation inside it can be considered as an ideal gas of photons with internal energy per unit volume$E=$ $\frac{U}{V} \propto {T^4}$ and pressure $P = \frac{1}{3}\left( {\frac{U}{V}} \right)$ If the shell now undergoes an adiabatic expansion the relation between $T$ and $R$ isView Solution
- 4The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$View Solution
- 5View SolutionA container that suits the occurrence of an isothermal process should be made of
- 6Two identical samples of a gas are allowed to expand $(i)$ isothermally $(ii)$ adiabatically. Work done isView Solution
- 7A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 8One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
- 9View SolutionThe given figure represents two isobaric processes for the same mass of an ideal gas, then
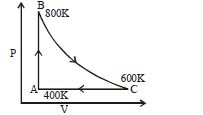
- 10One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution