The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$
Medium
(c) $P{V^\gamma } = K$or $P\gamma \,{V^{\gamma - 1}}dV + dP.\;{V^\gamma } = 0$
or $\frac{{dP}}{P} = - \gamma \frac{{dV}}{V}$ or $\frac{{dP}}{P} \times 100 = - \gamma \,\left( {\frac{{dV}}{V} \times 100} \right)$
$ = - 1.4 \times 5 = 7\% $
or $\frac{{dP}}{P} = - \gamma \frac{{dV}}{V}$ or $\frac{{dP}}{P} \times 100 = - \gamma \,\left( {\frac{{dV}}{V} \times 100} \right)$
$ = - 1.4 \times 5 = 7\% $
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If heat is added at constant volume, $6300\,\,J$ of heat are required to raise the temperature of an ideal gas by $150\,\,K$. If instead, heat is added at constant pressure, $8800$ joules are required for the same temperature change. When the temperature of the gas changes by $300\,\,K$, the internal energy of the gas changes by ..... $J$View Solution
- 2Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 3$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 4View SolutionIn the figure shown, amount of heat supplied to one mole of an ideal gas is plotted on the horizontal axis and amount of work done by gas is drawn on vertical axis. Assuming process be isobaric i.e. gas can be
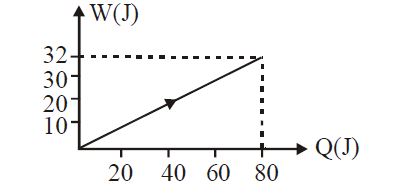
- 5A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution
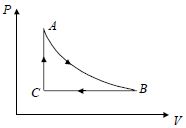
- 6For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
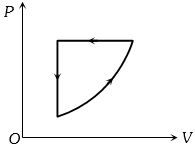
- 7View SolutionCofficient of performance of refigerator is
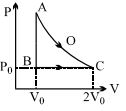
- 8An ideal gas is taken from point $A$ to point $C$ on $P-V$ diagram through two process $AOC$ and $ABC$ as shown in the figure. Process $AOC$ is isothermalView Solution
- 9$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
- 10A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution