A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$
JEE MAIN 2022, Medium
$T _{ c }=324 \; k$
$T _{ H }= ?$
$Q _{ H }=300 \; J$
$Q _{ L }=180 \; J$
$1-\frac{ Q _{ L }}{ Q _{ H }}=1-\frac{ T _{ L }}{ T _{ H }}$
$\frac{ Q _{ L }}{ Q _{ H }}=\frac{ T _{ L }}{ T _{ H }}$
$T _{ H }=\frac{ Q _{ H }}{ Q _{ L }} \times T _{ L }=\frac{300}{180} \times 324=540 K$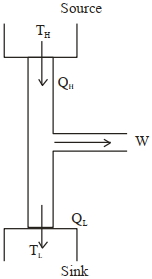
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
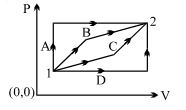
- 1An ideal gas is taken from state $1$ to state $2$ through optional path $A, B, C \& D$ as shown in $P-V$ diagram. Let $Q, W$ and $U$ represent the heat supplied, work done $\&$ internal energy of the gas respectively. ThenView Solution
- 2$Assertion :$ Air quickly leaking out of a balloon becomes cooler.View Solution
$Reason :$ The leaking air undergoes adiabatic expansion. - 3An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .View Solution
- 4An ideal gas follows a process described by $p V^2=C$ from $\left(p_1, V_1, T_1\right)$ to $\left(p_2, V_2, T_2\right)$ and $C$ is a constant. Then,View Solution
- 5View SolutionWhich of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
- 6For a refrigerator, heat absorbed from source is $800\, J$ and heat supplied to sink is $500\, J$ then the coefficient of performance isView Solution
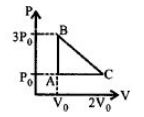
- 7One mole of an ideal monoatomic gas is taken along the path $ABCA$ as shown in the $PV$ diagram. The maximum temperature attained by the gas along the path $BC$ is given byView Solution
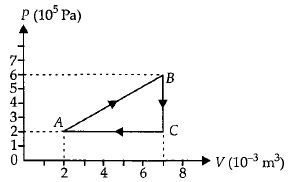
- 8A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
- 9How much work to be done in decreasing the volume of and ideal gas by an amount of $2.4 \times {10^{ - 4}}{m^3}$ at normal temperature and constant normal pressure of .......$joule$ $1 \times {10^5}N/{m^2}$View Solution
- 10When an ideal gas in a cylinder was compressed isothermally by a piston, the work done on the gas was found to be $1.5 \times {10^4}\;joules$. During this process aboutView Solution
