An iron rod of heat capacity $C$ is heated to temperature $8T_0$ . It is then put in a cylindrical vessel of adiabatic walls having two moles of air which can be treated as diatomic ideal gas at temperature $T_0$ and closed by a movable piston which is also adiabatic. The atmospheric pressure is $P_0$ . The cylinder with the piston combined have heat capacity $2C$ . Find the equilibrium temperature . (Assume temperature of air to be uniform and equal to vessel at all times) .
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
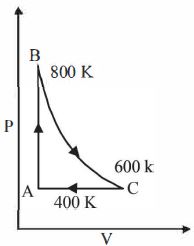
- 1One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statementView Solution
- 2View SolutionIn the adiabatic compression, the decrease in volume is associated with
- 3An ideal gas has volume ${V_0}$ at ${27^o}C.$ It is heated at constant pressure so that its volume becomes $2{V_0}.$ The final temperature isView Solution
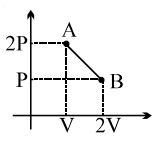
- 4The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 5View SolutionThe first operation involved in a Carnot cycle is
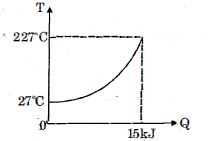
- 6$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
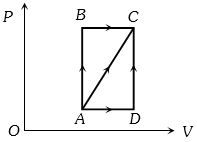
- 7A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 8$\Delta U + \Delta W = 0$ is valid forView Solution
- 9View SolutionIn an isothermal expansion
- 10An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
