$p V^2=C \Rightarrow\left(\frac{n R T}{V}\right) \cdot V^2=C$
$[\therefore$ for an ideal gas, $p V=n R T]$
$\Rightarrow \quad T V=\frac{C}{n R}=$ a constant
$\therefore \quad T \propto-\frac{1}{V}$
So, if $V_2 > V_1$, then $T_2 < T_1$.
Download our appand get started for free
Similar Questions
- 1One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
- 2View SolutionIn an isothermal process the volume of an ideal gas is halved. One can say that
- 3The efficiency of Carnot's engine operating between reservoirs, maintained at temperatures ${27^o}C$ and $ - {123^o}C,$ is ...... $\%$View Solution
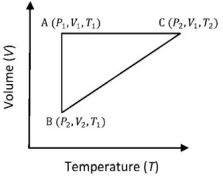
- 4A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
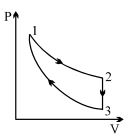
- 5Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 6View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 7Given below are two statements. One is labelled as Assertion A and the other is labelled as Reason $R$.View Solution
Assertion $A$ : If $dQ$ and $dW$ represent the heat supplied to the system and the work done on the system respectively. Then according to the first law of thermodynamics $d Q=d U-d W$.
Reason $R :$ First law of thermodynamics is based on law of conservation of energy.
In the light of the above statements, choose the correct answer from the option given below :
- 8In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by aboutView Solution
- 9Work done by $0.1$ mole of a gas at ${27^o}C$ to double its volume at constant pressure is ....... $cal$ ($R = 2 \,cal\, mol^{-1}$ $^oC^{-1}$)View Solution
- 10A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
